
Summary
To allow the combustion and destruction efficiencies of a flare system to be estimated, the molecular weight of the flare gas can be helpful. This is normally derived from the composition of the flare gas.
If taking a sample for laboratory analysis or on-line compositional analysis is not possible, then, if the density of the gas that will be routed to flare is known along with its pressure and temperature, the molecular weight can be calculated. In very limited circumstances this molecular weight enables the composition mixture of the flare gas to be estimated.
How it Works
Using the ideal gas law, we know that at low pressures the density of a gas is directly proportional to its molecular weight as follows:
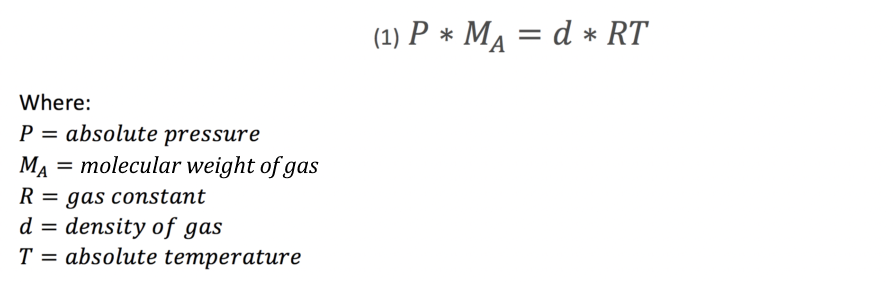
Equation (1) is valid for a single component of gas, to derive for a two gas mixture, we require the following calculation:
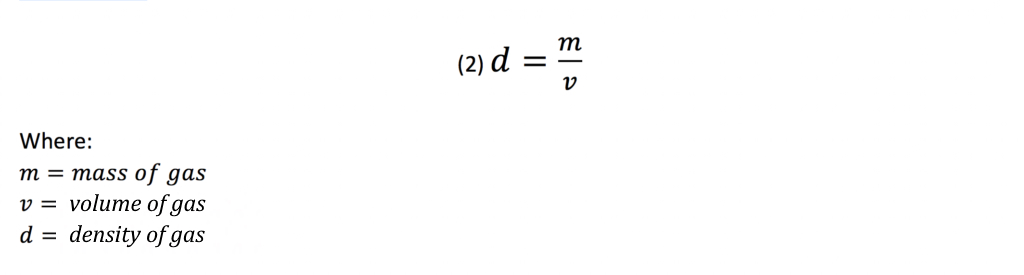
The density of a mixture of two gases A and B could be obtained from the total mass of the gases as follows:

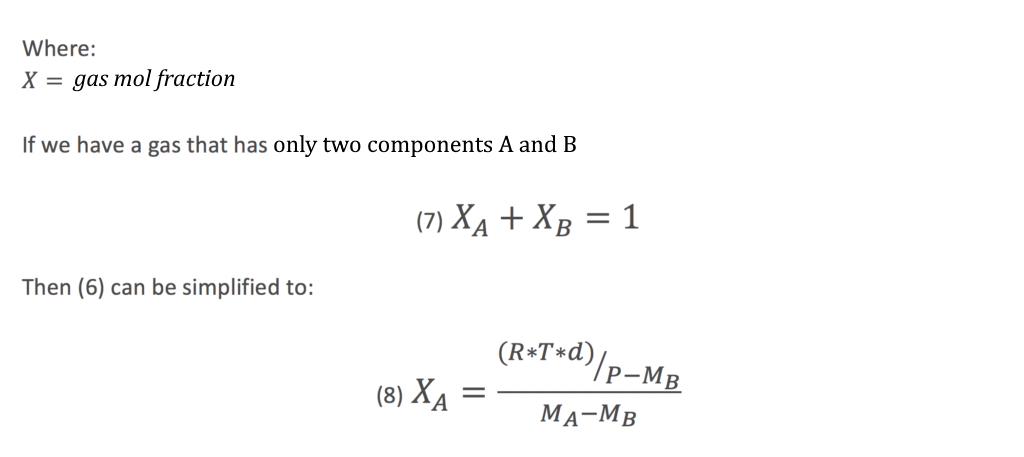
From equation (8) using the measured density of the flare gas, we can derive the components of the composition of the flare gas providing we know which two components it is likely to contain.
Equation (8) can then be entered into either the flare meter controller/flow computer or within the DCS / ICSS to derive the composition of the gas, the corresponding molecular weight which will enable the calculation of the combustion and destruction efficiency.
Utilisation of the inference of composition from measured density is not a common application, but can be used when no sampling or analysis facilities are available on the flare systems.
Advantages
No Equipment costs
Minimal Maintenance costs
On-line continuous calculation
Limitations
Only applicable for flare gas containing no more than two known compounds
Low Accuracy
High Uncertainty
Case study
No case study available at this time.
On-line analytical methods involve extracting a sample from a flare system via an automatic sampling system and analysing it continuously with a field installed analyser system.